Research

Explore breakthroughs and ongoing studies in lung cancer research, from clinical trials to cutting-edge therapies and prevention.
Learn about local and national events where you can connect, raise awareness, and support the lung cancer community.
Explore real stories honoring survivors and those remembered through their lasting legacy. Select a category to find stories that resonate most with you.
Find practical advice, emotional support, and resources to help you or your loved one cope with the challenges of living with lung cancer.
Stay informed about the policies that impact the lung cancer community and ways you can get involved.
Explore breakthroughs and ongoing studies in lung cancer research, from clinical trials to cutting-edge therapies and prevention.
Catch up on highlights from our newsletter, featuring stories, resources, and upcoming opportunities to engage.
Explore news and updates shaping the lung cancer landscape—from scientific breakthroughs and organizational milestones to awareness campaigns and community highlights.


May 5, 2026

For Amita Jain, MD, lung cancer is not just a diagnosis. It’s a lived experience that has shaped her life across multiple roles as a physician, a patient, a daughter, a caregiver, and an advocate.
Her family’s lung cancer story spans generations. In July 2018, her mother, Usha Jain, a retired UC Berkeley professor, was diagnosed with stage 4 (IV) non-small cell lung cancer (NSCLC) despite having no smoking history. Just 6 months later, Amita herself would receive the same diagnosis.
While both women faced advanced disease, their treatment paths diverged. Usha underwent chemotherapy and immunotherapy for nearly 2 years before stopping treatment and transitioning to routine scans. That period (often described clinically as “watch and wait”) felt anything but passive to her family.
“It was hard to feel as though we were ‘doing nothing,’” Amita recalls.
Turning to navigation for answers
As both a physician and a caregiver, Amita understood medicine, but even she found the complexity of lung cancer care overwhelming.
Navigating next steps after treatment, understanding emerging options, and keeping up with rapidly evolving research can feel like a full-time job, especially for families already carrying the emotional weight of a diagnosis.
So, Amita reached out to GO2's LungMATCH navigation program.
“I called the navigators and furnished my mom’s tumor’s genetic profile,” she says. “They reached out a day later with information on some trials for which she might qualify.”
Although her mother ultimately chose not to pursue clinical trials, the impact of that interaction was profound.
“While we did not take action on the options that the navigator provided, the information was empowering,” Amita explains. “It really helped us understand the ‘lay of the land’ in her particular situation.”
Bridging the gap between information and understanding
Even for someone with medical training, the experience revealed an important truth. Access to information is not the same as understanding it.
“Navigating is complex and scary, and getting all the information is difficult if you are not an oncologist,” Amita says. “The navigator that I spoke with was both professional and prompt. That information made me feel as though we understood our options—and that was extremely reassuring.”
For patients and families, that reassurance can be transformative.
A lung cancer diagnosis often brings not only fear, but also a sense of powerlessness. Decisions feel urgent; stakes are high, and the volume of information can be paralyzing.
“The navigator can serve as a bridge to knowledge,” Amita says. “Knowledge is power. Having a diagnosis of lung cancer can be shocking, but more importantly you do feel a bit powerless and overwhelmed. The navigators serve as a resource and lifeline.”
The invisible work of navigation
What many patients don’t see is the depth of work happening behind the scenes.
Navigators are constantly reviewing evolving research, identifying clinical trials, interpreting biomarker data, and translating complex medical information into something patients can actually use to discuss with their care teams.
“Keeping up on the information is a full-time job that most of us are not trained to do,” Amita says.
That expertise becomes especially critical at moments of uncertainty, like when treatment ends, and the path forward is unclear.
In those moments, navigation doesn’t just guide decisions; it restores a sense of control.
Strengthening navigation for the future
Stories like Amita’s mother’s underscore the critical role navigators play, not just in coordinating care, but in empowering patients and families with clarity, confidence, and connection.
To support and strengthen this role, GO2 for Lung Cancer partnered with leading experts to develop a comprehensive resource for those on the front lines of patient care.
The Lung Cancer Navigator: A Guide for Nurses and Allied Health Professionals textbook is designed to equip health professionals with the knowledge and tools they need to guide patients through every step of the lung cancer experience, from diagnosis to survivorship.
By investing in navigator education, we can ensure that more patients and families experience what Amita describes so clearly: the shift from feeling overwhelmed and uncertain to being informed and empowered.

April 17, 2026
The absence of tobacco use suggests alternative carcinogenic mechanisms may contribute to lung cancer development in younger individuals.
SAN CARLOS, Calif., April 17, 2026 — The Addario Lung Cancer Medical Institute (ALCMI) and GO2 for Lung Cancer today announced results from a new study presented at the 2026 American Association for Cancer Research annual meeting showing that lifestyle patterns and dietary exposures could play a role in tumor mutations among young adults with lung cancer. The results, drawn from the ongoing Epidemiology of Young Lung Cancer study, may help uncover modifiable risk factors in a population where lifestyle behaviors traditionally associated with lung cancer, such as smoking, fail to explain disease onset.
This project is conducted by researchers from California Northstate University College of Medicine, the Dana-Farber Cancer Institute, The Ohio State University Comprehensive Cancer Center, and USC Norris Comprehensive Cancer Center, part of Keck Medicine of USC. The work is supported by AstraZeneca, the Beth Longwell Foundation, Genentech, GO2 for Lung Cancer, and Upstage Lung Cancer.
There is growing concern that lung cancer is biologically distinct in adults under 40, with 84% of patients carrying targetable mutations. These historically younger patients are predominantly female and have no smoking history, which raises questions about other potential environmental contributors.
The new analysis examined data from 187 patients, assessing dietary patterns and food exposure in conjunction with mutation subtypes. Key findings include:
- Diet quality and pesticide-linked foods. Patients with EGFR and fusion mutations reported better overall diet quality than the U.S. population average. However, their higher intake of fruits, vegetables, and whole grains, which are often associated with elevated pesticide residue, may represent an overlooked environmental risk factor.
- Unexpected tobacco associations. In the fusion pathway group (ALK, ROS1, RET, NTRK), 55.6% were people with no smoking history, challenging conventional links between these mutations and tobacco exposure. Conversely, one-third of EGFR pathway patients reported a smoking history, higher than anticipated.
- Potential connection to oral contraceptives. Oral contraceptive use was consistently elevated across all biological pathway groups. Although most patients reported 1-5 years of use, the median duration was 10 years due to a subset of long-term users. This matters because the pesticides can act as endocrine disruptors that interact with estrogen receptors seen in the lung cancers most prevalent in the young lung cancer population.
The study team notes that further research is needed to clarify how dietary pesticide exposure interacts with genetic pathways and whether reducing exposure could lower risk.
“These findings demonstrate that there is still so much to learn about this terrible disease, particularly as it occurs in young people,” said ALCMI Executive Director and COO Richard Erwin. “Our hope is that this is just the beginning of this research and that others will join us in exploring how and why lung cancer is presenting in the younger patient population.”
“This work represents a critical step toward identifying modifiable environmental factors that may contribute to lung cancer in younger adults,” said Jorge J. Nieva, MD, a thoracic oncologist with USC Norris Comprehensive Cancer Center. “Our hope is that these insights can guide both public health recommendations and future research into prevention.”
“Too many young people diagnosed with lung cancer are left asking, ‘how did this happen?’ especially when they have no known risk factors,” said Chief Patient Officer and Co-Interim CEO of GO2 for Lung Cancer Danielle Hicks. “This research reflects what patients have long suspected. There may be risk factors beyond their control. It’s a critical step toward understanding risk, improving early detection, and giving patients clearer answers.”
These findings would not be possible without the vision of ALCMI Co-Founder Bonnie J. Addario, who passed away in 2025. She long suspected an unknown environmental link could be contributing to this disease and was steadfast in her determination to search for answers for lung cancer patients.
“While more research is needed to confirm these findings, this is the first clear evidence that lung cancer in younger patients may develop through a different biological pathway,” said Tony Addario, ALCMI co-founder and husband of the late Bonnie Addario. “This discovery is both scientific and deeply personal. Bonnie’s vision has guided this research from the very beginning, and her legacy continues in the hope we bring to every person facing lung cancer, and in the lives we may one day prevent from ever being touched by it.”
Disclosure:
Dr. Nieva has received consulting payments from AstraZeneca and Genentech.

April 14, 2026
Small cell lung cancer (SCLC) is 1 of the 2 main types of lung cancer and accounts for about 15 out of 100 (15%) cases. For decades, treatment for SCLC included chemotherapy, radiation therapy, or surgery if diagnosed in early stages.
It wasn’t until 2018 that the first immunotherapy for SCLC was approved by the Food and Drug Administration (FDA). This was an important advancement in treating SCLC, as immunotherapy works very differently from other treatment types. In October 2025, another breakthrough occurred in the field of immunotherapy when a new type of immunotherapy, a T-cell engager, was approved for SCLC.
These advancements bring hope and optimism to SCLC and expand treatment options. Understanding immunotherapy, how it works, and when it may be used helps you have informed conversations with your healthcare team and stay involved in decision-making. This is important because being knowledgeable allows you to advocate for what matters most to you.
What is immunotherapy?
Your immune system defends your body against germs and diseases that can cause illness. Most of the time, it can tell which cells are healthy and which are not. This enables the immune system to target disease and germ cells without harming healthy cells.
When your immune system detects cancer cells, it goes to work destroying them. Sometimes, it has trouble finding cancer cells because they can appear like healthy cells. Immunotherapy helps your immune system find and attack cancer cells.
Types of immunotherapy
Currently, there are 2 main types of immunotherapies used in SCLC: checkpoint inhibitors and T-cell engagers. They each work in very different ways and are used in different points in treatment.
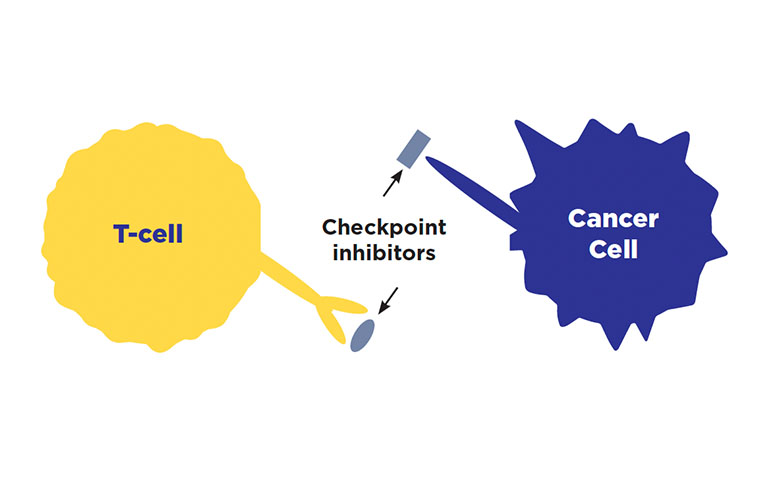
Checkpoint inhibitors
How do they work?
Cancer cells can hide from the immune system by attaching themselves to immune cells. Cancer cells can attach to immune cells at an area called a checkpoint. This sends a signal to your immune system that the cancer cell is healthy, which prevents the immune system cell from attacking it. A checkpoint inhibitor works by blocking cancer cells from attaching to immune system cells, allowing the immune system to better attack the cancer.

When are they used to treat SCLC?
Imfinzi (durvalumab) and Tecentriq (atezolizumab); however, recent clinical trials have broadened their approved uses in different ways.
- Limited-stage SCLC (stages 1, 2, 3): Imfinzi (durvalumab) can be used after chemotherapy and radiation treatments are complete and may be continued for up to 2 years.
- Extensive-stage SCLC (stage 4): Imfinzi (durvalumab) or Tecentriq (atezolizumab) can be used in combination with chemotherapy. Imfinzi may be continued alone after chemotherapy is completed for as long as it is effective and well-tolerated. This is called maintenance therapy. Tecentriq (atezolizumab) may also be used as maintenance therapy either alone or combined with the chemotherapy drug Lurbinectedin.
T-cell engagers
How do they work?
T-cell engagers, such as Imdelltra (tarlatamab), work like a matchmaker. They attach to a T-cell (a type of immune cell that fights germs and diseases) and a cancer cell, bringing them close together. This connection helps your immune system find and attack cancer cells.
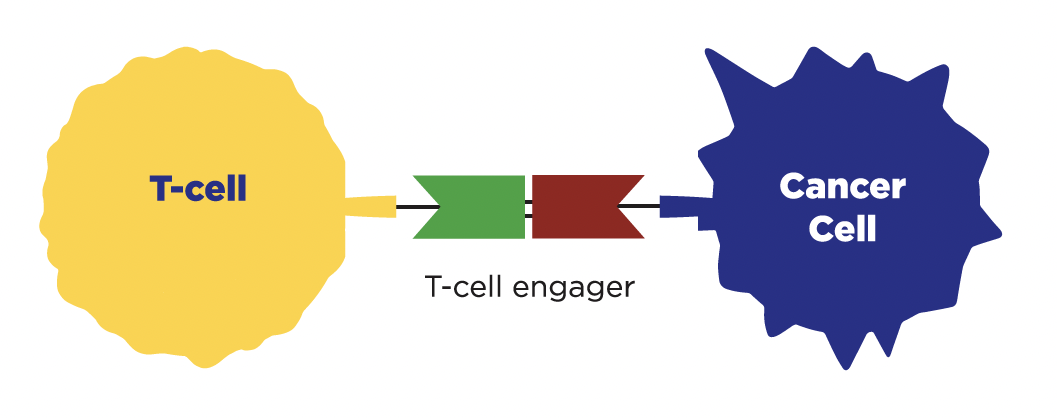
When are they used to treat SCLC?
T-cell engagers are FDA-approved for use in extensive-stage SCLC that has spread during or after chemotherapy.
Immunotherapy side effects
The side effects you may experience from immunotherapy will be unique to you. Some common side effects are mild, while others can be more severe. Immunotherapy boosts your immune system's activity, which can sometimes impact healthy cells and organs, leading to inflammation. This requires prompt medical attention.
Ask a member of your healthcare team what common and more serious side effects may occur with your specific type of immunotherapy and know when you should seek immediate medical care.
The future of immunotherapy in SCLC
Immunotherapy is not 1 single treatment; it’s a growing toolbox of therapies. In fact, there are many clinical trials currently underway that focus on:
- Expanding how the currently approved immunotherapies are used to improve outcomes, including using them in earlier stages or combined with other treatments
- Developing new immunotherapy treatments that work differently from the current options
- Exploring ways to boost the body’s immune system and impact the tumor to make it easier to target and attack Questions for your healthcare team
It’s important to talk to your healthcare team about all your treatment options, including immunotherapy. The following questions may be helpful to ask in your conversation:
- Is immunotherapy right for me?
- What side effects should I be aware of with the type of immunotherapy recommended for me?
- Are there any clinical trials that may be right for me?


March 30, 2026
By the time many people with cancer begin treatment, the barriers they face, including confusing medical information, fragmented care, transportation challenges, and financial strain, have often been building for weeks or even months.
This is why patient navigation programs were created. Navigators help patients schedule appointments, understand treatment options, coordinate care among multiple providers, and connect with resources that address financial or logistical barriers. For people facing complex healthcare systems, navigation can make the difference between timely treatment and dangerous delays.
But there is a question we rarely ask: Are these programs being designed around the needs of the people most affected by cancer disparities?
A recent scoping review conducted by researchers at GO2 for Lung Cancer and published in an ASCO journal examined cancer navigation studies across the United States and Canada. The goal was simple: to understand what kinds of support patients say they need and whose voices are shaping the research guiding these programs.
The findings revealed several consistent challenges. Across the studies reviewed, patients most often reported difficulties with communication with their healthcare providers, emotional and psychological support following diagnosis, and basic access to care. Many patients described struggling to understand complex medical information, coordinate appointments across multiple specialists, or manage the emotional toll of a cancer diagnosis.
Yet the most striking finding was not the needs themselves. It was who was missing from the research.
Despite experiencing some of the highest cancer mortality rates in North America, African American and American Indian or Alaska Native populations were rarely represented in the studies reviewed. Only a small portion of the research included these groups, even though they face well-documented barriers to screening, diagnosis, and treatment.
At the same time, many studies failed to report key factors that strongly influence cancer care experiences, including whether patients lived in rural or urban areas or whether they faced financial hardship. Without this context, it becomes difficult to fully understand the barriers patients encounter while navigating cancer care.
The timing of these findings is critical. Health systems across North America are increasingly investing in patient navigation programs to reduce disparities and improve outcomes. But if the research guiding these programs does not reflect the communities most affected by cancer disparities, there is a risk that well-intentioned solutions will miss the barriers that matter most.
Some critics may argue that expanding navigation programs should be the priority, regardless of who is represented in the research. Navigation programs have indeed shown tremendous promise in improving care coordination and helping patients move through complex healthcare systems.
But expanding programs without ensuring that the evidence reflects the experiences of the most vulnerable populations at risk, reinforcing the very disparities these programs are meant to solve.
Cancer care does not always require complicated solutions. Sometimes the first step is simpler: making sure the people most affected by the problem are the ones guiding the conversation about the solution.
Until cancer navigation research consistently reflects the voices of the communities most affected by cancer disparities, the healthcare system will continue trying to solve problems without fully understanding them.


February 18, 2026
On February 6th, 2026, the U.S. Food and Drug Administration (FDA) approved Median Technologies’ eyonis Lung Cancer Screening (LCS), a medical device used for the combined detection and diagnosis of lung cancer. eyonis LCS is an AI-based technology designed to support the earlier detection of lung cancer by helping clinicians identify and characterize pulmonary (lung) nodules on low-dose CT (LDCT) scans.
In performance testing, eyonis LCS demonstrated a:
- 93.3% sensitivity, which means that if 100 people actually have lung cancer, the device will correctly identify about 93 of them. In other words, the device reliably detects cancer when it is present.
- 92.4% specificity, which means that if 100 people don’t have lung cancer, the device will correctly identify about 92 of them as cancer-free. In other words, the device largely avoids false alarms.
- 99.9% negative predictive value, which means that if the device says you don’t have cancer, there’s a 99.9% chance you truly don’t have it. In other words, a negative result is highly reliable.
These outcomes demonstrate the powerful potential for eyonis LCS to be used as a tool to enhance lung screening programs by improving screening accuracy, while reducing unnecessary follow-up procedures. The ability to detect suspicious pulmonary nodules earlier may also help clinicians diagnose lung cancer at earlier stages more frequently, where patient outcomes can be significantly improved.
If you or a loved one is eligible for lung cancer screening, ask your healthcare team about eyonis LCS. For questions about lung cancer treatments, trials, or biomarker testing, contact our LungMATCH team at support@go2.org or 1-800-298-2436.
Read the FDA approval.


February 3, 2026
By Matthew Reiss, MSE, PhD, Manager, Precision Medicine & Navigation, GO2 for Lung Cancer
Hearing that cancer has spread to the brain or spinal cord can be a frightening, but very real outcome for many people living with lung cancer. While exact statistics are hard to pin down, recent studies suggest that about 10-40% of lung cancer patients’ cancer will spread to the brain or central nervous system.
One rare form of spread, called leptomeningeal disease (LMD), can be especially confusing because it is unfamiliar and often hard to explain in simple terms. But better understanding what LMD is and how it may be treated can help you advocate for yourself in conversations with your healthcare team.
What is leptomeningeal disease?
To understand leptomeningeal disease, it can help to picture how the brain and spinal cord are protected. In the body, these structures are wrapped in a thin layer of tissue called the meninges. Inside these layers flows a clear liquid called cerebrospinal fluid (CSF), which helps to cushion and nourish the brain and spinal cord.
Leptomeningeal disease occurs when cancer cells, such as those from the lungs, travel to, and start growing in, the meninges or in the CSF surrounding the brain and spinal cord.
How is LMD different from other brain metastases?
Many people have heard how cancer can sometimes spread from the lungs by travelling through the bloodstream to the brain, where it can begin to grow. These are called brain metastases or brain mets. Brain mets are not brain cancer, but rather lung cancer that has spread to the brain.
Leptomeningeal disease is different. While brain mets usually form as a solid tumor(s) in the brain itself, LMD involves the lining and fluid around the brain and spinal cord. Instead of a single or multiple solid tumor masses, LMD can look more like a thin coating or a pattern of streaking around/within these structures, possibly affecting many areas at once. This can make LMD harder to see on a scan and more challenging to diagnose
Because of this, symptoms of LMD can vary widely, and no 2 people experience LMD the same way. Often, symptoms include:
- Headaches
- Nausea
- Changes in thinking or mood
- Double vision
- Weakness
- Difficulty walking
- Back pain
- Numbness
This also makes LMD more difficult to diagnose. Often, an LMD diagnosis is confirmed by collectively considering the results of MRI scans, spinal fluid tests, and other clinical exams.
How common is LMD in lung cancer?
Leptomeningeal disease is not common, but it is a possibility to be aware of for individuals living with lung cancer.
For individuals with non-small cell lung cancer (NSCLC), LMD is very rare:
- Studies show that LMD occurs in about 3-5% of individuals living with NSCLC.
- It is more common in people with mutations or changes in genes such as EGFR, ALK, and ROS1.
LMD is more common, but still rare, in people living with small cell lung cancer (SCLC):
- About 2% of individuals with SCLC have signs of LMD at diagnosis.
- About 10% of individuals with SCLC have developed LMD after 2 years.
You may also sometimes hear people say that LMD diagnoses are becoming more common. While true, many researchers believe LMD is being diagnosed more often today, not only because of better scanning technology, but also because new treatments are allowing people to live long enough with lung cancer for rarer complications like LMD to occur.
Even though it’s uncommon, knowing the basics of LMD can help people better recognize symptoms early and get connected with a specialist.
The importance of seeing a specialist
Because LMD is complex, it is very helpful to work with clinicians who have experience treating it. Your doctor may refer you to a:
- Neuro-oncologist – a doctor specializing in cancers found in the brain and nervous system
- Medical oncologist with LMD experience
- Radiation oncologist – a doctor specializing in the use of radiation to treat cancer in areas such as the brain and spine
These experts work with your current healthcare team and can often open the door to more treatment options or clinical trials to help manage the cancer.
Treating LMD can also be complex. Sometimes treatments that work well in one part of the body do not work the same way or as effectively in the brain or CSF. If this happens to you or your loved one, it’s completely normal to feel overwhelmed. You can always ask your care team to slow down, repeat information, or clarify anything. Asking questions is an important way to advocate for yourself.
Current treatment approaches for LMD
Overall, the goals of treatment for people with LMD are usually to slow or control further spread of the cancer, protect quality of life, and relieve troublesome symptoms such as pain, nausea, or nerve issues.
Exactly which treatment someone receives for LMD is highly personalized, but there are several commonly used approaches:
- Targeted or systemic (whole-body) treatments: For some people who have certain genetic changes, targeted therapies or immunotherapies can be effective ways to treat LMD. For others who do not have genetic changes that can be targeted, traditional chemotherapy is often used.
- Radiation therapy: Targeted radiation may also be used to treat the specific areas of LMD that are likely causing symptoms. For others, radiation may be used more widely along the brain or spinal cord to treat LMD over a large region.
- Intrathecal therapy: In some cases, medicines such as chemotherapy are delivered directly into the spinal fluid through a small device or spinal tap. This approach allows the treatment to better access the protected part of the body where LMD is occurring, although this approach is not appropriate for everyone.
- Supportive and palliative care: This approach includes medicines and services that focus on symptom relief, mobility, and emotional support for people with LMD, rather than treating the LMD directly. Supportive and palliative care should be offered alongside other treatment approaches.
- Emerging approaches: While LMD remains a serious diagnosis, there is real progress being made in clinical trials to find new and better ways to treat LMD and improve long-term outcomes. Talk to your healthcare team about whether a clinical trial for LMD may be right for you.
Closing thoughts
Leptomeningeal disease is complicated, and it is normal to have questions. If you or someone you care about has been told they have leptomeningeal disease related to lung cancer, reach out to your care team, ask about a referral to an expert, and feel comfortable asking questions related to your treatment options. Clear information is empowering and can help you advocate for the best possible care.
If you’d like to learn more about LMD and its treatments, have questions, or need support, please contact our free HelpLine at 1-800-298-2436 or email support@go2.org.


January 5, 2026
By Matthew Reiss, MSE, PhD, Manager, Precision Medicine & Navigation, GO2 for Lung Cancer
This is the third blog post of our ongoing “Understanding Lung Cancer” series. This series aims to simplify the science behind lung cancer and help provide a deeper understanding of the disease. In the first and second parts, we introduced the basics of cells and explored how DNA damage can lead to cancer. We also discussed what drives cancer to grow and spread. This time, we’ll take a closer look at the role your immune system plays in cancer. We will also tackle how cancer cells avoid your immune system, and introduce the major types of lung cancer treatments used today, including approaches that are shaping the future of care.
Your immune system: the body’s defense team
Aside from the cell’s own means of identifying abnormal cells and preventing them from growing, your body has another line of defense: your immune system. The immune system is made up of cells, tissues, and organs that work together. It protects you from infections, repairs injuries, and removes cells that are damaged or no longer needed. One of its most important jobs is to identify and destroy cells that aren’t acting normally, including cells that could become cancer. Think of it like an army inside your body. It is always on the lookout for anything that doesn’t belong, ready to fight it off to keep you healthy.
All cells have small “markers” or proteins found on their surface that tell the immune system whether they are healthy or not. If a cell seems abnormal, the immune system will target and destroy the cell before it becomes a bigger problem.
So you may wonder: If the immune system can remove normal cells, why doesn’t it always stop cancer?
How cancer cells avoid the immune system
Cancer cells are clever. As they grow and change, they learn new ways to hide from or block the immune system. There are several ways cancer cells do this, but the most common ways include:
- Hiding their abnormal markers so that immune cells can’t recognize them
- Displaying markers on their surfaces that act as “don’t attack me” messages to immune cells
- Creating an environment around the cancer cells that weakens the immune response
- Growing so quickly that the immune system can’t keep up
Cancer cells may use one or more of these methods to continue growing, even when the immune system tries to fight back.
Applying what we’ve learned
Now that we understand how cancer cells grow and how they avoid the immune system, we can focus on how current treatments work to address these challenges.
In addition to surgery, which is a treatment where a surgeon aims to remove the cancer itself, there are several commonly used treatment approaches in lung cancer.
Chemotherapy: a long-standing tool in cancer care
Chemotherapy, or “chemo,” works by attacking a cell’s ability to divide. It is known as a “systemic treatment,” meaning it travels through the bloodstream to affect the whole body, not just a specific, localized area. Unfortunately, this means that chemotherapy does not distinguish between healthy cells and cancer cells, which is why chemo often affects fast-growing cells throughout the body, including hair and blood cells. As a result, common side effects include hair loss and low blood cell counts.
Because cancer cells divide so much faster than healthy cells, chemotherapy can still be a useful treatment option for many people. Improvements over the years have made many chemo treatments easier to tolerate.
Radiation therapy: using energy to kill cancer cells
Radiation therapy uses high-energy beams to damage the DNA inside cells, including cancer cells. This slows their growth or destroys them. It can be delivered from outside the body, aimed directly at the tumor. In some cases, radiation is given directly to the tumor inside the body itself.
Because radiation has the potential to also affect healthy cells, it’s usually given in specialized ways that minimize its effect on healthy cells while directing most of the radiation to the cancer cells specifically. As a result, radiation usually affects fewer healthy cells than chemotherapy, and today’s techniques are more precise than ever before.
Targeted therapy: targeting what makes cancer cells different
We’ve previously talked about driver mutations. These are specific changes in the DNA of cancer cells that cause them to grow. Targeted therapies are treatments specifically designed to attack cells containing these driver mutations. Healthy cells are often spared, so people generally have fewer side effects.
The same targeted therapy won’t work for everyone, because not everyone will have a driver mutation that can be treated using targeted therapies. This is why biomarker testing is so important, specifically in non-small cell lung cancer (NSCLC). The targeted therapy used is matched to each person’s test results.
There are many different kinds of targeted therapies, and new ones are being developed every day, but the 2 most commonly used targeted therapies in lung cancer are:
- Tyrosine kinase inhibitors (TKIs), which work by blocking the activity of the driver mutation in cancer cells to slow down or stop their growth.
- Antibody-drug conjugates (ADCs) that link targeting antibodies to chemotherapy-like drugs to deliver treatments directly to cancer cells. Think of it as “targeted chemo”.
Immunotherapy: using your immune system to fight back
Immunotherapies (sometimes called “IO”) are treatments that help the immune system recognize and attack cancer cells more effectively. Currently, immunotherapies fall into 2 main classes:
- Checkpoint inhibitors are a type of cancer treatment that blocks the cancer cells’ ability to hide from your immune system. This allows your immune system to better find and attack the cancer.
- T-cell engagers are a type of cancer treatment that helps T-cells (a type of immune cell) better connect to cancer cells. This connection helps the immune system attack the cancer more effectively.
New treatments on the horizon
Cancer research moves quickly. New treatments, including those that fall into the categories above, are always in development. But before they are used more widely, new treatments must first be studied in clinical trials to ensure they are safe and effective. Clinical trials give lung cancer patients a chance to try promising new treatments. They also help doctors learn what works best for future care.
Everyone is different. The exact treatment your healthcare team recommends may include one or more of the treatments listed above. It’s important to speak with your healthcare team about which treatment is right for you.
Coming up next: lung cancer risks and prevention
In the final part of this series, we’ll explore the factors that can affect lung cancer risk. These include genetics, environment, and lifestyle choices. We’ll also discuss what individuals and communities can do to support prevention, early diagnosis, and better access to care.


December 18, 2025
On December 17, 2025, the U.S. Food and Drug Administration (FDA) approved Rybrevant Faspro (amivantamab and hyaluronidase-lpuj) for subcutaneous injection, commonly known as a shot. This approval included all the same uses of Rybrevant (amivantamab) that the FDA previously approved for adults with metastatic non-small cell lung cancer (NSCLC) whose tumors have an EGFR mutation.
This latest approval is based on the results of the PALOMA-3 study, which compared the use of Rybrevant Faspro (amivantamab and hyaluronidase-lpuj) given by a shot (injection) under the skin versus traditional Rybrevant (amivantamab), which is given as an infusion into the bloodstream (intravenous/IV). People given Rybrevant Faspro (amivantamab and hyaluronidase-lpuj) as a shot received the drug over approximately 5 minutes compared to the standard IV, which can sometimes take a few hours.
The study showed both administration methods to be equally effective, and individuals receiving Rybrevant Faspro (amivantamab and hyaluronidase-lpuj) by injection experienced significantly fewer administration-related reactions. This latest approval offers greatly improved patient convenience in receiving treatment and lowers the burden of healthcare resources needed.
Please speak with your healthcare team for more information about Rybrevant Faspro (amivantamab and hyaluronidase-lpuj) to see if it may be a good option for you. If you have questions about treatment, trials, or biomarker testing, contact our LungMATCH team at support@go2.org or 1-800-298-2436.
Read the full FDA announcement.


December 1, 2025
Spend just a few minutes talking with people who have been diagnosed with lung cancer, and a familiar rhythm begins to emerge: “I never thought I’d be diagnosed with lung cancer.” “I eat healthy… I run and lift weights… I’ve never even smoked.” Then come the stories—the unexpected turns that changed everything. A scan after a car accident revealed a suspicious nodule. A runner noticed she was getting winded on her usual route. A lingering cough led to a test, and then another, until the unthinkable became real. Each story is different, yet the same question echoes through them all: Why me?
While doctors focus on finding the best ways to treat lung cancer, our researchers are looking deeper into the environments, lifestyles, and histories that might help explain why lung cancer develops in some people and not in others.
We’re asking questions like:
- What kinds of environments, at home or at work, have people spent the most time in?
- What does their health history look like?
- What about diet and exercise habits?
To explore these questions, GO2 for Lung Cancer created a planning committee made up of people living with lung cancer, along with doctors and professionals who treat and study it. Together, we reflected on one clear truth: the earlier lung cancer is discovered, the better the chance for effective treatment and survival
From those discussions came a new initiative: A set of surveys designed to learn both from people living with lung cancer and those who don’t have it but may have experienced certain risk factors. By comparing responses, we hope to identify patterns that could lead to earlier detection, and ultimately, save more lives through prevention and diagnosis.
The Lung Cancer Risk Survey is a 4-part questionnaire that asks about your health, lifestyle, and the environments where you’ve spent the most time. It takes about 20–25 minutes to complete.
Who can take the survey?
You may qualify if any of the following apply:
- You use or used tobacco products
- You have a family history of lung cancer
- You’ve worked in jobs with poor air quality (military, firefighting, mining, etc.)
- You’ve been exposed to radon
- You have lung diseases like COPD or pulmonary fibrosis
- You’ve lived through major air pollution events (like 9/11 or wildfires)
Jeffery Velotta, MD, FACS, at Kaiser Permanente Oakland Medical Center, has been an early supporter of GO2’s Lung Cancer Risk Survey.
“I love how it really looks into other significant risk factors beyond traditional smoking and pack years—because as we know, those other risk factors are crucial in the development of lung cancer. They can be additive and perhaps even more applicable, especially for younger people who get lung cancer,” said Dr. Velotta.
If you’ve spent time working in smoke-related environments, responded to disasters like 9/11 or wildfires, mined underground, or simply lived in areas with poor air quality—your lived experience could make a difference. You can help researchers learn how factors at home, at work, and in the environment may affect lung health.
Please join us in shaping the future of lung cancer research. If you qualify, take the survey, share it with others, and help us uncover patterns behind lung cancer risk. Your feedback can guide researchers, potentially leading to earlier detection and saving lives. Learn more.
