Blog
Whether you're navigating a diagnosis or supporting someone who is, our blog is here to inform, inspire, and connect. Discover stories of hope, expert insights, and the latest updates in lung cancer care—all in one place.

Use the search box or filter buttons above to find a blog that interests you.
Learn about local and national events where you can connect, raise awareness, and support the lung cancer community.
Explore real stories honoring survivors and those remembered through their lasting legacy. Select a category to find stories that resonate most with you.
Find practical advice, emotional support, and resources to help you or your loved one cope with the challenges of living with lung cancer.
Stay informed about the policies that impact the lung cancer community and ways you can get involved.
Explore breakthroughs and ongoing studies in lung cancer research, from clinical trials to cutting-edge therapies and prevention.
Catch up on highlights from our newsletter, featuring stories, resources, and upcoming opportunities to engage.
Explore news and updates shaping the lung cancer landscape—from scientific breakthroughs and organizational milestones to awareness campaigns and community highlights.


May 28, 2026
WASHINGTON, D.C., May 27, 2026 — GO2 for Lung Cancer (GO2) is proud to announce Jennifer W. Li, MD, of Johns Hopkins University, as the recipient of the 2026 Conquer Cancer – GO2 for Lung Cancer Young Investigator Award in memory of Debbie Consiglio.
Presented in collaboration with Conquer Cancer®, the ASCO Foundation, the Young Investigator Award (YIA) supports promising early-career physicians during the final years of training as they transition to faculty appointments and pursue innovative clinical oncology research. The mentored award supports physician-scientists conducting research under the guidance of experienced scientific mentors and is designed to help investigators transition from fellowship training into independent academic careers. Applications are accepted from investigators across all areas of cancer research from both the United States and around the world.
Through this one-year grant, Dr. Li will lead a research project titled, “Integrative blood-based proteomic and circulating tumor DNA dynamics for early immunotherapy response and toxicity detection in advanced lung cancer.” Her work aims to improve the ability to predict which patients will benefit from immunotherapy and identify those at risk for serious treatment-related side effects using minimally invasive blood-based testing.
Immunotherapy has transformed treatment for many people living with lung cancer, yet significant challenges remain. While some patients experience durable responses, others do not benefit, and approximately 10–20% develop severe side effects that can significantly impact quality of life. Dr. Li’s research seeks to combine analyses of protein and circulating tumor DNA collected before and during treatment to better understand treatment response and toxicity in real time.
“GO2 is proud to support emerging researchers whose work has the potential to directly improve the lives of people living with lung cancer,” said GO2’s Co-Interim Chief Executive Officer and Chief Patient Officer Danielle Hicks. “Dr. Li’s research reflects the kind of patient-centered innovation that is urgently needed to help patients and their care teams make more informed treatment decisions, better manage side effects, and ultimately improve outcomes. Through our longstanding collaboration with Conquer Cancer, we remain committed to advancing research that brings more personalized and compassionate care to the lung cancer community.”
Dr. Li is a medical oncology fellow at the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins University, where her research focuses on precision oncology and biomarker development in lung cancer. Her work integrates multi-omic and liquid biopsy approaches to improve the prediction of treatment response, toxicity, and clinical outcomes. She plans to pursue a career as an academic thoracic oncologist.
“I am incredibly honored to receive this award,” said Dr. Li. “This support will help advance our work to better understand why patients respond differently to immunotherapy and why some develop severe side effects. My hope is that this research will contribute to more personalized, safer, and more effective treatment strategies for people living with lung cancer.”
Dr. Li studied biological engineering at the Massachusetts Institute of Technology, where she worked on microfluidic platforms for single-cell analysis to support individualized cancer care. She earned her medical degree from the University of Florida and completed her internal medicine residency at Brown University. Her current research combines molecular profiling, liquid biopsy, and data-driven approaches to advance personalized cancer treatment.
GO2 proudly supports Conquer Cancer’s Grants & Awards Program and the next generation of lung cancer researchers. Through this collaboration, grants have funded innovative studies exploring topics such as radiation response, biomarker detection, and novel treatment strategies. The program reflects GO2’s commitment to encouraging emerging investigators to pursue careers dedicated to improving outcomes for people impacted by lung cancer.
Conquer Cancer will formally recognize this year’s award recipients during the 2026 ASCO Annual Meeting.
The 2026 Conquer Cancer – GO2 for Lung Cancer Young Investigator Award in memory of Debbie Consiglio is supported by Natera.


May 13, 2026
A diagnosis of small cell lung cancer (SCLC) can bring a wide range of emotions. If you’re feeling uncertain, afraid, or overwhelmed, connecting with others who have faced similar experiences can offer both comfort and strength. Hearing from people who have been living well with SCLC for years is a powerful reminder that your story is still unfolding.
Connection can take many forms, including support groups, one-on-one peer support, or simply reading others’ stories. Each of these forms of connection can help ease feelings of isolation and provide a sense of shared understanding. You are not alone in this. Others are walking this path too, and when asked, here’s what they want you to know.
Take it one step at a time
Focus on today - this appointment, this decision, this moment. Thinking too far ahead can be overwhelming when there is so much unknown in the path ahead. Try to stay in the present and remember that progress isn’t about having everything figured out. It’s about continuing forward one step at a time. There will be good days and hard days. Both are part of the process.
Advocate for yourself
Your healthcare team matters, and you are the most important person on that team. Your thoughts, feelings, and perspectives guide your treatment and care decisions. Your healthcare team should listen and respect you. Whether it’s asking questions, exploring clinical trials, or discussing needs, your voice matters.
- Ask questions until you understand your diagnosis and treatment options.
- Consider getting a second opinion from a thoracic oncologist (lung cancer specialist).
- If something doesn’t feel right, trust your instincts and consider a change.
Let people in (even when it’s hard)
Support can make a huge difference, but accepting help isn’t always easy. The offer of help from friends and family is one way they show their love for you. Accepting their help allows them to be with you through this experience and has the power to strengthen relationships and deepen bonds.
Take care of your body and keep moving
Self-care, even on tough days, can help you both physically and mentally. Even small actions matter.
- Spend time in nature
- Go for a walk when you can
- Eat and hydrate as best you’re able
- Follow your care team’s guidance
- Do something you enjoy
Be honest about how you feel and focus on what you can control
Positivity helps, but it doesn’t mean ignoring fear, frustration, or sadness. It’s okay to feel all of it. What matters is not getting stuck there. Give yourself permission to have hard moments, then try to find your way forward, step by step.
There’s a lot you can’t control with cancer. But there are things you can:
- How you spend your time
- Who you surround yourself with
- How you approach decisions
Some people find strength in learning everything they can. Others focus on daily routines or small goals. There’s no single right way, only what works for you.
Find meaning where you can and hold onto hope
Many people describe a shift in perspective after diagnosis. Time feels different. Priorities become clearer. That means something different for everyone. For you, it could mean:
- Spending more time with loved ones
- Helping others facing cancer
- Finding purpose in advocacy or community
- Setting goals or challenges for yourself and meeting them
Discover what brings you peace, laughter, and enjoyment, and then do more of that.
SCLC is serious. It’s okay to acknowledge that. But hope has a definite place here, not as denial, but as possibility. Treatments are improving. Research is ongoing. And your experience is your own. You don’t have to have everything figured out. You just keep moving forward, one step, one decision, one day at a time.
If you would like to learn more about SCLC, have questions, or need support, our SCLC program can help. Contact our free HelpLine at 1-800-298-2436 or email support@go2.org.


May 6, 2026

Michelle Bos-Lun is a third term state representative in the Vermont General Assembly. She lives in Westminster with her husband. She has 3 adult children and 2 grandchildren. Michelle is an enthusiastic vegan cook and baker, a mushroom forager, a hiker, and an advocate for lung cancer.
When Michelle returned home from a trip to Nepal last year, she soon found herself unable to get out of bed. At first, she thought she had jet lag, but when her symptoms worsened, she ended up in the hospital.
What followed was a whirlwind of tests and uncertainty that led to a diagnosis she hadn’t expected: scrub typhus, a rare and potentially life-threatening infection she had contracted while traveling.
But the story didn’t end there. In the process of trying to understand why she was so sick, doctors ordered a chest X-ray and saw something there that they couldn’t explain.
Three weeks and even more tests later, Michelle received a second surprising diagnosis: lung cancer.
A diagnosis she never expected
Like so many others, lung cancer wasn’t something Michelle imagined would happen to her until it did.
“Before my diagnosis, I knew of one person who had lung cancer without a smoking history,” she said. “So, while I knew it was possible, it didn’t seem at all likely. But lots of people aren’t good candidates for lung cancer, and yet it still finds us.”
Still, even in the face of a stage 3B (IIIB) diagnosis, her reaction was not what many might expect.
“I’m pretty pragmatic about things,” she said. “When something comes up, I want to learn all about it and figure out what I can do to have the best results with a situation that isn’t ideal.”
That instinct to learn, adapt, and move forward has shaped everything that’s followed

“I won the lottery”
After her initial diagnosis, Michelle entered a waiting period, one filled with a lot of questions, but few clear answers. Her oncologist couldn’t yet tell her what kind of lung cancer she had or how it would be treated. The outlook he shared at that point was sobering.
But there was one 1 possibility that offered hope, and that was a genetic mutation.
Her doctor told her, “If you have a mutation, there are some really good targeted therapies.”
Two weeks later, the call came. Michelle had ALK-positive lung cancer.
“He told me I won the lottery,” she said. “I had the best one with the best treatment.”
Within days of starting the targeted therapy, Lorbrena (loralatinib), her persistent cough, which was her only noticeable symptom, had disappeared. A few months later, her first scan showed her cancer had been reduced by nearly 50%.
“My doctor actually said, ‘This is even better than I would have expected,’” Michelle said.
A whole-person approach
From the beginning, Michelle approached her diagnosis the same way she has approached much of her life by integrating different perspectives, experiences, and tools. Having spent part of her adolescence in Taiwan, and as a long-practicing Buddhist, she was already comfortable holding multiple ways of thinking at once.
“My life has been a mixture of East and West since I was 12,” she said. “So, when I got cancer, it made sense to include a variety of different resources and approaches.”
Her treatment plan reflects that. Alongside her targeted therapy, Michelle has built a daily routine that supports her body in other ways:
- Tai Chi twice a day to improve circulation and manage neuropathy
- Walking several miles each day
- Bi-weekly acupuncture
- Regular lymphatic massage
- A nutrient-dense, plant-based diet including daily kale smoothies, large salads, and roasted vegetables with whole grains
“I feel as healthy as I’ve ever been. Except I have cancer,” she said.
It’s a paradox she doesn’t shy away from. And it’s one she’s actively working to sustain.
“The lorlatinib gets credit for 75% of my healing, and my kale smoothies get the other 25%,” she joked.
Choosing hope—on purpose
Michelle is quick to acknowledge that everyone responds to a diagnosis differently. But for her, hope is not something she has to force; it’s something she naturally gravitates toward.
“I think you can choose to look at the hardest parts, or you can look at the possible good outcomes,” she said. “For me, it’s more satisfying to look at the positive. I try to find the best-case scenarios and aim for those.”
Even before her diagnosis was confirmed, she made a conscious choice not to dwell in pessimism.
“Before my lung biopsy, I thought, I don’t think it’s very likely that this is lung cancer, so I’m not going to worry about it,” she said. “And then when it turned out to be cancer, I said, ‘Okay, so when can we figure out how to treat this?’ I could have spent that time scared, but being scared wouldn’t have changed the outcome, only how I experienced the time leading up to it. That’s sort of how I approach everything.”
Finding community and perspective
Like many people diagnosed with a biomarker-driven form of lung cancer, Michelle quickly found community among others living with ALK-positive disease.
“I feel like I’ve been welcomed into this club that none of us wanted to join,” she said. “But since we’re here, we’re there for each other.”
Through that community, she found not only information, but perspective, including examples of people living 10, 15, even 20 years after diagnosis. Their stories helped her understand the full range of what’s possible and where she might fit within it.
“Somebody would say, ‘my husband is 21 years out’, and I thought, what? We can live 21 years with this? Knowing that some people can live quite a long time made me feel like, okay, then I can make it too,” she said.

Not waiting anymore
If there is 1 theme that runs through Michelle’s story, it’s this: don’t wait.
After her diagnosis, she began thinking about the people she hadn’t seen in years and the friendships that had drifted with time and distance. She immediately set out to rectify that. Her first trip was to reconnect with a close friend she hadn’t seen in decades.
“I just thought, I don’t want to wait,” she said. “Why did I already wait 30 years? I’m not doing that again.”
Now, her “bucket list” isn’t about places; it’s about people – reunions, visits, and time spent together.
“I hope I’m going to be around for a long time,” she said. “But in case I’m not, I don’t want to wait to spend time with the people who matter most to me. I want to see them now.”
Turning experience into action
As a state legislator in Vermont, Michelle already had a platform. After her diagnosis, she chose to use it in a new way. From the very beginning, she has spoken publicly about her experiences, using them as an opportunity to raise awareness about lung cancer, and especially the importance of early detection.
“I thought, maybe I can help somebody else,” she said. “If I can help educate someone, then go ahead, use my story.”
After her story appeared on the front page of her local paper, she learned that at least two people went to their doctors to ask for chest X-rays. For Michelle, that was enough.
“That’s the whole point,” she said.
She is also working to connect her personal experience with broader public health efforts, including initiatives that emphasize nutrition as a core component of care.
“When people eat healthy food, it helps keep them healthy,” she said. “I’m a living example of that.”
Moving forward
Less than a year into her diagnosis, Michelle’s life looks both familiar and entirely new. She is still working, still advocating, and still showing up for her family.
But she is also living with a deeper sense of urgency and intention.
“When you get lemons, make lemonade,” she said. “Well, I’m making some lemonade.”
Only in Michelle’s case, it’s likely organic, and paired with a kale smoothie.
A lung cancer diagnosis can bring a lot of questions, emotions, and uncertainty. GO2 for Lung Cancer’s free HelpLine connects you with trained specialists who can provide support, answer questions, and help you find resources along the way. Whether you’re newly diagnosed, in treatment, or navigating survivorship, you don’t have to face it alone.
Call us at 1-800-298-2436 or email support@go2.org to learn more.


May 6, 2026

For Janaki and Harsha, their wedding wasn’t just about celebrating a new chapter together; it was also an opportunity to honor the past, give back, and make a meaningful impact for others facing lung cancer.
Janaki’s connection to lung cancer is deeply personal. Her late husband, Kartik, was diagnosed with stage 4 (IV) lung cancer at just 28 years old while he was still a third-year internal medicine resident. Through a targeted clinical trial for his EML4/ALK mutation, he was able to continue working for several years before passing in 2015 at the age of 34. His journey continues to inspire Janaki’s commitment to supporting others impacted by the disease.
This was a second marriage that brought together 2 families, not starting from scratch. Material gifts didn’t feel necessary, but they knew their loved ones would still want to give something. That’s when they decided to turn their wedding into a fundraiser in honor of Kartik. “Many of our friends and family were familiar with Kartik’s journey,” Janaki said. “They were more than happy to donate, and several told us they thought it was a great idea.”
Lessons learned along the way
While their fundraiser was a success, Janaki reflects on what she might do differently.
“We didn’t initially go through GO2 because we were trying to get donations matched through companies,” she explained. “But in retrospect, it would have been easier, and likely more effective, to set up a GO2 fundraising page and link it directly on our wedding website.”
Planning ahead, she noted, could have streamlined the process and avoided fees from other fundraising platforms.
But what surprised them most was the incredible generosity of their community. “We were amazed by how much money we were able to raise.”

Tips for turning your own wedding into a fundraiser
For couples considering a similar approach, Janaki offers practical advice:
- Start early. Wedding planning gets busy, so give yourself time to set things up properly.
- Keep it simple. A direct link to a fundraising page makes it easy for guests to contribute.
- Spread the word beyond the website. Share your fundraiser at pre-wedding events or gatherings. Many guests won’t check every page on a website.
- Create in-person opportunities. If you’re hosting events leading up to the wedding, like bridal showers, engagement parties, etc, consider setting up a donation box.
- Ask for help. Friends and family are often eager to support both your wedding and your cause.
Continuing the impact
Even beyond their fundraiser, Janaki continues to connect others to GO2’s resources and recently referred a family friend for support.
“I love seeing what the organization is doing for patients and in research,” she said. “It’s providing hope and that’s one of the most important things when facing lung cancer.”
Make your celebration count
Inspired by Janaki and Harsha’s story? You can turn your own milestone—whether it’s a wedding, birthday, or special event—into a powerful way to support people impacted by lung cancer.
Start your own fundraiser benefiting GO2 and help bring hope to patients and families who need it most.
Learn more about how you can create your own fundraiser for GO2 for Lung Cancer.


May 5, 2026

For Amita Jain, MD, lung cancer is not just a diagnosis. It’s a lived experience that has shaped her life across multiple roles as a physician, a patient, a daughter, a caregiver, and an advocate.
Her family’s lung cancer story spans generations. In July 2018, her mother, Usha Jain, a retired UC Berkeley professor, was diagnosed with stage 4 (IV) non-small cell lung cancer (NSCLC) despite having no smoking history. Just 6 months later, Amita herself would receive the same diagnosis.
While both women faced advanced disease, their treatment paths diverged. Usha underwent chemotherapy and immunotherapy for nearly 2 years before stopping treatment and transitioning to routine scans. That period (often described clinically as “watch and wait”) felt anything but passive to her family.
“It was hard to feel as though we were ‘doing nothing,’” Amita recalls.
Turning to navigation for answers
As both a physician and a caregiver, Amita understood medicine, but even she found the complexity of lung cancer care overwhelming.
Navigating next steps after treatment, understanding emerging options, and keeping up with rapidly evolving research can feel like a full-time job, especially for families already carrying the emotional weight of a diagnosis.
So, Amita reached out to GO2's LungMATCH navigation program.
“I called the navigators and furnished my mom’s tumor’s genetic profile,” she says. “They reached out a day later with information on some trials for which she might qualify.”
Although her mother ultimately chose not to pursue clinical trials, the impact of that interaction was profound.
“While we did not take action on the options that the navigator provided, the information was empowering,” Amita explains. “It really helped us understand the ‘lay of the land’ in her particular situation.”
Bridging the gap between information and understanding
Even for someone with medical training, the experience revealed an important truth. Access to information is not the same as understanding it.
“Navigating is complex and scary, and getting all the information is difficult if you are not an oncologist,” Amita says. “The navigator that I spoke with was both professional and prompt. That information made me feel as though we understood our options—and that was extremely reassuring.”
For patients and families, that reassurance can be transformative.
A lung cancer diagnosis often brings not only fear, but also a sense of powerlessness. Decisions feel urgent; stakes are high, and the volume of information can be paralyzing.
“The navigator can serve as a bridge to knowledge,” Amita says. “Knowledge is power. Having a diagnosis of lung cancer can be shocking, but more importantly you do feel a bit powerless and overwhelmed. The navigators serve as a resource and lifeline.”
The invisible work of navigation
What many patients don’t see is the depth of work happening behind the scenes.
Navigators are constantly reviewing evolving research, identifying clinical trials, interpreting biomarker data, and translating complex medical information into something patients can actually use to discuss with their care teams.
“Keeping up on the information is a full-time job that most of us are not trained to do,” Amita says.
That expertise becomes especially critical at moments of uncertainty, like when treatment ends, and the path forward is unclear.
In those moments, navigation doesn’t just guide decisions; it restores a sense of control.
Strengthening navigation for the future
Stories like Amita’s mother’s underscore the critical role navigators play, not just in coordinating care, but in empowering patients and families with clarity, confidence, and connection.
To support and strengthen this role, GO2 for Lung Cancer partnered with leading experts to develop a comprehensive resource for those on the front lines of patient care.
The Lung Cancer Navigator: A Guide for Nurses and Allied Health Professionals textbook is designed to equip health professionals with the knowledge and tools they need to guide patients through every step of the lung cancer experience, from diagnosis to survivorship.
By investing in navigator education, we can ensure that more patients and families experience what Amita describes so clearly: the shift from feeling overwhelmed and uncertain to being informed and empowered.


April 21, 2026
GO2 for Lung Cancer today applauds House passage of H.R. 2319, the Women and Lung Cancer Research and Preventive Services Act, an important bipartisan step toward final enactment this Congress.
We commend the leadership of Representatives Brendan Boyle (D- 2- PA) and Brian Fitzpatrick (R-1-PA). We also thank Speaker Mike Johnson (R-4-LA), Majority Leader Steve Scalise (R-1-LA), and Democratic Leader Hakeem Jeffries (D-8-NY) for advancing the bill under suspension and recognize the leadership of the House Energy and Commerce Committee, including Chairman Brett Guthrie (R-2-KY) and Ranking Member Frank Pallone (D-6-NJ).
This milestone builds on more than a decade of advocacy and research, including work with the Connors Center for Women’s Health and Gender Biology at Brigham and Women's Hospital, which first identified critical gaps in understanding lung cancer in women and called for a coordinated national response.
While smoking rates have declined, lung cancer diagnoses, particularly among women, have not kept pace. This underscores a critical truth that lung cancer can affect anyone, including people with no known risk factors. Yet this misconception continues to drive stigma and delay diagnosis. The key unanswered question is why younger, otherwise healthy women with no traditional risk factors are developing lung cancer.
“The placement of this bill on the House Suspension Calendar reflects strong bipartisan recognition that we must do more to address lung cancer in women,” said GO2 for Lung Cancer’s Interim Co-CEO Danielle Hicks. “Once implemented, this legislation will improve understanding of genetic, hormonal, environmental, and behavioral factors and strengthen prevention, screening, and treatment strategies for everyone impacted by lung cancer.”
“The advancement of this bill is about more than research funding, it is about correcting a dangerous and outdated narrative,” said GO2 for Lung Cancer’s Senior Director of Government Affairs, Elridge Proctor. “We are still seeing lung cancer in younger women with no known risk factors. That means critical scientific gaps remain, and patients are paying the price.”
By advancing a more complete understanding of disease risk beyond risk factors, the bill helps dismantle stigma at its root, replacing blame with evidence and ensuring that all patients are recognized, studied, and served.
GO2 for Lung Cancer urges the Senate to swiftly pass S. 1157 and conference with the House on the two versions to bring the bill to the President’s desk. We stand ready to work with federal agencies and leading patient advocacy organizations to ensure this legislation is implemented and delivers meaningful progress for everyone living with lung cancer.
Join us in taking action. Contact your Senator, every message counts!

April 17, 2026
The absence of tobacco use suggests alternative carcinogenic mechanisms may contribute to lung cancer development in younger individuals.
SAN CARLOS, Calif., April 17, 2026 — The Addario Lung Cancer Medical Institute (ALCMI) and GO2 for Lung Cancer today announced results from a new study presented at the 2026 American Association for Cancer Research annual meeting showing that lifestyle patterns and dietary exposures could play a role in tumor mutations among young adults with lung cancer. The results, drawn from the ongoing Epidemiology of Young Lung Cancer study, may help uncover modifiable risk factors in a population where lifestyle behaviors traditionally associated with lung cancer, such as smoking, fail to explain disease onset.
This project is conducted by researchers from California Northstate University College of Medicine, the Dana-Farber Cancer Institute, The Ohio State University Comprehensive Cancer Center, and USC Norris Comprehensive Cancer Center, part of Keck Medicine of USC. The work is supported by AstraZeneca, the Beth Longwell Foundation, Genentech, GO2 for Lung Cancer, and Upstage Lung Cancer.
There is growing concern that lung cancer is biologically distinct in adults under 40, with 84% of patients carrying targetable mutations. These historically younger patients are predominantly female and have no smoking history, which raises questions about other potential environmental contributors.
The new analysis examined data from 187 patients, assessing dietary patterns and food exposure in conjunction with mutation subtypes. Key findings include:
- Diet quality and pesticide-linked foods. Patients with EGFR and fusion mutations reported better overall diet quality than the U.S. population average. However, their higher intake of fruits, vegetables, and whole grains, which are often associated with elevated pesticide residue, may represent an overlooked environmental risk factor.
- Unexpected tobacco associations. In the fusion pathway group (ALK, ROS1, RET, NTRK), 55.6% were people with no smoking history, challenging conventional links between these mutations and tobacco exposure. Conversely, one-third of EGFR pathway patients reported a smoking history, higher than anticipated.
- Potential connection to oral contraceptives. Oral contraceptive use was consistently elevated across all biological pathway groups. Although most patients reported 1-5 years of use, the median duration was 10 years due to a subset of long-term users. This matters because the pesticides can act as endocrine disruptors that interact with estrogen receptors seen in the lung cancers most prevalent in the young lung cancer population.
The study team notes that further research is needed to clarify how dietary pesticide exposure interacts with genetic pathways and whether reducing exposure could lower risk.
“These findings demonstrate that there is still so much to learn about this terrible disease, particularly as it occurs in young people,” said ALCMI Executive Director and COO Richard Erwin. “Our hope is that this is just the beginning of this research and that others will join us in exploring how and why lung cancer is presenting in the younger patient population.”
“This work represents a critical step toward identifying modifiable environmental factors that may contribute to lung cancer in younger adults,” said Jorge J. Nieva, MD, a thoracic oncologist with USC Norris Comprehensive Cancer Center. “Our hope is that these insights can guide both public health recommendations and future research into prevention.”
“Too many young people diagnosed with lung cancer are left asking, ‘how did this happen?’ especially when they have no known risk factors,” said Chief Patient Officer and Co-Interim CEO of GO2 for Lung Cancer Danielle Hicks. “This research reflects what patients have long suspected. There may be risk factors beyond their control. It’s a critical step toward understanding risk, improving early detection, and giving patients clearer answers.”
These findings would not be possible without the vision of ALCMI Co-Founder Bonnie J. Addario, who passed away in 2025. She long suspected an unknown environmental link could be contributing to this disease and was steadfast in her determination to search for answers for lung cancer patients.
“While more research is needed to confirm these findings, this is the first clear evidence that lung cancer in younger patients may develop through a different biological pathway,” said Tony Addario, ALCMI co-founder and husband of the late Bonnie Addario. “This discovery is both scientific and deeply personal. Bonnie’s vision has guided this research from the very beginning, and her legacy continues in the hope we bring to every person facing lung cancer, and in the lives we may one day prevent from ever being touched by it.”
Disclosure:
Dr. Nieva has received consulting payments from AstraZeneca and Genentech.

April 14, 2026
Small cell lung cancer (SCLC) is 1 of the 2 main types of lung cancer and accounts for about 15 out of 100 (15%) cases. For decades, treatment for SCLC included chemotherapy, radiation therapy, or surgery if diagnosed in early stages.
It wasn’t until 2018 that the first immunotherapy for SCLC was approved by the Food and Drug Administration (FDA). This was an important advancement in treating SCLC, as immunotherapy works very differently from other treatment types. In October 2025, another breakthrough occurred in the field of immunotherapy when a new type of immunotherapy, a T-cell engager, was approved for SCLC.
These advancements bring hope and optimism to SCLC and expand treatment options. Understanding immunotherapy, how it works, and when it may be used helps you have informed conversations with your healthcare team and stay involved in decision-making. This is important because being knowledgeable allows you to advocate for what matters most to you.
What is immunotherapy?
Your immune system defends your body against germs and diseases that can cause illness. Most of the time, it can tell which cells are healthy and which are not. This enables the immune system to target disease and germ cells without harming healthy cells.
When your immune system detects cancer cells, it goes to work destroying them. Sometimes, it has trouble finding cancer cells because they can appear like healthy cells. Immunotherapy helps your immune system find and attack cancer cells.
Types of immunotherapy
Currently, there are 2 main types of immunotherapies used in SCLC: checkpoint inhibitors and T-cell engagers. They each work in very different ways and are used in different points in treatment.
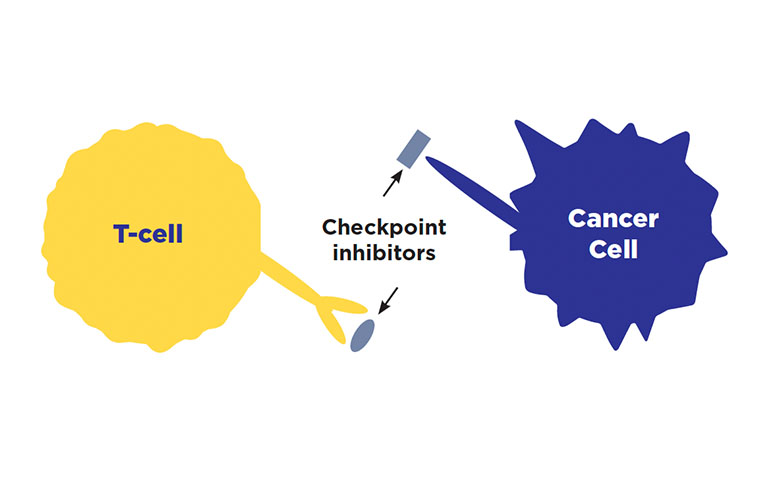
Checkpoint inhibitors
How do they work?
Cancer cells can hide from the immune system by attaching themselves to immune cells. Cancer cells can attach to immune cells at an area called a checkpoint. This sends a signal to your immune system that the cancer cell is healthy, which prevents the immune system cell from attacking it. A checkpoint inhibitor works by blocking cancer cells from attaching to immune system cells, allowing the immune system to better attack the cancer.

When are they used to treat SCLC?
Imfinzi (durvalumab) and Tecentriq (atezolizumab); however, recent clinical trials have broadened their approved uses in different ways.
- Limited-stage SCLC (stages 1, 2, 3): Imfinzi (durvalumab) can be used after chemotherapy and radiation treatments are complete and may be continued for up to 2 years.
- Extensive-stage SCLC (stage 4): Imfinzi (durvalumab) or Tecentriq (atezolizumab) can be used in combination with chemotherapy. Imfinzi may be continued alone after chemotherapy is completed for as long as it is effective and well-tolerated. This is called maintenance therapy. Tecentriq (atezolizumab) may also be used as maintenance therapy either alone or combined with the chemotherapy drug Lurbinectedin.
T-cell engagers
How do they work?
T-cell engagers, such as Imdelltra (tarlatamab), work like a matchmaker. They attach to a T-cell (a type of immune cell that fights germs and diseases) and a cancer cell, bringing them close together. This connection helps your immune system find and attack cancer cells.
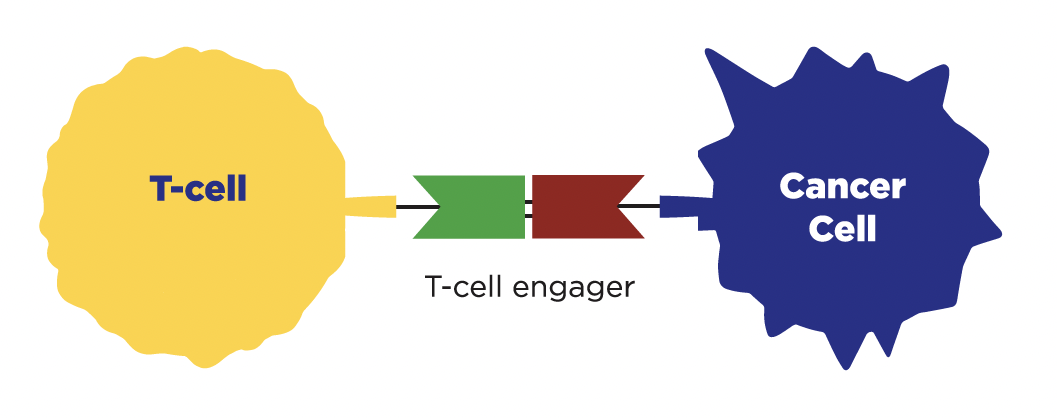
When are they used to treat SCLC?
T-cell engagers are FDA-approved for use in extensive-stage SCLC that has spread during or after chemotherapy.
Immunotherapy side effects
The side effects you may experience from immunotherapy will be unique to you. Some common side effects are mild, while others can be more severe. Immunotherapy boosts your immune system's activity, which can sometimes impact healthy cells and organs, leading to inflammation. This requires prompt medical attention.
Ask a member of your healthcare team what common and more serious side effects may occur with your specific type of immunotherapy and know when you should seek immediate medical care.
The future of immunotherapy in SCLC
Immunotherapy is not 1 single treatment; it’s a growing toolbox of therapies. In fact, there are many clinical trials currently underway that focus on:
- Expanding how the currently approved immunotherapies are used to improve outcomes, including using them in earlier stages or combined with other treatments
- Developing new immunotherapy treatments that work differently from the current options
- Exploring ways to boost the body’s immune system and impact the tumor to make it easier to target and attack Questions for your healthcare team
It’s important to talk to your healthcare team about all your treatment options, including immunotherapy. The following questions may be helpful to ask in your conversation:
- Is immunotherapy right for me?
- What side effects should I be aware of with the type of immunotherapy recommended for me?
- Are there any clinical trials that may be right for me?


March 30, 2026

From March 1–3, 2026, lung cancer advocates, researchers, survivors, and partners from across the country gathered in Washington, D.C., for the annual GO2 for Lung Cancer Voices Summit. This year’s summit marked a milestone moment for the lung cancer community, bringing together passionate advocates to elevate patient voices and push for policies that accelerate research and improve outcomes for people affected by lung cancer.
GO2 was proud to partner with organizations and advocates across the full spectrum of the lung cancer community—biomarker groups, research foundations, advocacy organizations, and patient-led initiatives—to make our collective voice impossible to ignore.
This unprecedented collaboration not only amplifies our shared mission but also demonstrates the strength, resilience, and expertise driving progress in lung cancer today.
We are deeply grateful to the advocates who traveled from across the country to meet directly with lawmakers and share their experiences. Their voices help ensure that lung cancer remains a national priority and that progress continues for the millions of individuals and families impacted by this disease.
By the numbers
This year’s summit was a milestone event in many ways. Highlights include:
- 140 meetings with legislative offices on Capitol Hill
- 139 attendees
- 29 states represented
- 63,705 social media impressions and 4,915 engagements
Our key priorities
Advocates urged Congress to take action on several policy priorities that support the lung cancer community.
- Our request for Fiscal Year 2027
Advocates called on Congress to fully restore and increase funding for the Lung Cancer Research Program (LCRP) to $60 million within the Department of Defense’s Congressionally Directed Medical Research Program (CDMRP). This investment is critical to reversing the impact of the FY 2025 full-year continuing resolution, which:- Enacted a 57 percent reduction in overall CDMRP funding
- Eliminated dedicated funding for the LCRP in FY 2025
- Support for federal research funding
The lung cancer community also urged policymakers to support the highest possible funding levels for key federal medical research agencies, including:- National Institutes of Health (NIH)
- National Cancer Institute (NCI)
- Advanced Research Projects Agency for Health (ARPA-H)
- Food and Drug Administration (FDA)
Sustained investment across these agencies is essential to drive the scientific discoveries and innovative treatments that improve survival and quality of life for patients.
Keynote speaker highlights the promise of progress
GO2 for Lung Cancer was honored to welcome Christine M. Lovly, MD, PhD, FASCO, as the summit’s keynote speaker. Dr. Lovly is the division chief of thoracic medical oncology and a professor in the Department of Medical Oncology & Therapeutics Research at City of Hope, one of the largest and most advanced cancer research and treatment organizations in the United States.
In her remarks, Dr. Lovly highlighted the remarkable progress made in lung cancer research and treatment over the past two decades while emphasizing the continued need for sustained funding and strong advocacy. She also spoke about the promise of future breakthroughs and the power of collaboration across the lung cancer community.
Survivor spotlight: shining a light on lung cancer
A powerful moment of the summit featured lung cancer survivor and advocate Dusty Donaldson, who shared her personal experience with lung cancer and the impact advocacy can have on driving change.
Through the Shine a Light on Lung Cancer® initiative, the presentation highlighted the importance of elevating patient voices, honoring those affected by lung cancer, and inspiring action to advance research, support, and hope for everyone impacted by the disease.
Advocacy award recognition
At the Voices Summit, GO2 recognizes and celebrates individuals who have made a meaningful impact in the lung cancer community.
This year, Bruce Dunbar received the 2026 Lung Cancer Voices Summit Advocacy Leadership Award for Excellence in Community Engagement & Awareness for his outstanding commitment to advancing progress for people affected by lung cancer. A lung cancer survivor and volunteer state team leader, Bruce has been a dedicated advocate and an active member of the Voices Summit Planning & Advisory Committee.
Raise your voice year-round
Advocacy does not end when the Voices Summit concludes. The lung cancer community can continue to make an impact year-round by contacting lawmakers and supporting policies that accelerate research, expand access to care, and improve outcomes.
Every message, every meeting, and every story shared helps build the momentum needed to drive lasting change for people affected by lung cancer. Take action today.
Pictures from the 2026 Lung Cancer Voices Summit
Our professional photographer captured many special moments at this year’s Voices Summit. Please review our photo album from the event, and feel free to download, share or post your favorite image to your social media network. Don’t forget to tag us too:
Facebook: GO2 for Lung Cancer
Instagram: @go2forlungcancer
LinkedIn: GO2 for Lung Cancer
Thank you to our 2026 Lung Cancer Voices Summit sponsors
AstraZeneca, Merck, AbbVie, Daiicho-Sankyo, Genentech, Grail, Johnson & Johnson, Pfizer, Novocure, Regeneron, Lilly, Natera, Takeda Oncology
